Plastic Melting Points: A Complete Guide

Plastic melting points explained: charts, influencing factors, and implications for material selection and manufacturing processes.
Introduction
Plastic materials are used across virtually every manufacturing industry, including injection-molded consumer products as well as high-performance 3D printed and thermoformed components. Despite their widespread use, the thermal limits of plastics are often misunderstood. In practice, a plastic’s melting point and its response to heat play a decisive role in material selection, processing stability, and long-term part performance.
Unlike metals, plastics do not exhibit a single, universal melting behavior. Some polymers melt sharply at a defined temperature, while others soften gradually over a broader range. This is why terms such as melting point, glass transition temperature (Tg), and heat deflection temperature (HDT) are frequently confused, leading to improper processing settings, dimensional instability, or premature part failure.
This guide breaks down how different plastics behave under heat, the factors that influence melting point, and the importance of melting point in real-world applications such as material selection and processing temperature control.
What Is the Melting Point of Plastic?
The melting point of plastic is the temperature at which a plastic material transitions from a solid state into a molten or flowable state, allowing it to be reshaped.
Unlike metals, not all plastics have a single, clearly defined melting point. Some plastics melt sharply at a specific temperature, while others soften gradually over a temperature range. This behavior is determined by the polymer’s molecular structure.
To avoid confusion, it is important to first understand how plastics are structurally classified and how that structure determines their thermal behavior.
The table below summarizes the key differences between semi-crystalline thermoplastics, amorphous thermoplastics, and thermosets, including:
whether the material undergoes true melting, gradual softening, or no melting at all
which temperature parameter matters most (Tm, Tg, or decomposition temperature)
and how molecular structure affects processing behavior
Thermoplastic: Semi-Crystalline | Thermoplastic: Amorphous | Thermoset | |
|---|---|---|---|
Chain Structure | Ordered / Stable | Random / Disordered | Cross-linked |
Thermal Behavior | Sharp Melting | Gradual Softening | Irreversible Curing Decomposes at high heat. |
Key Temperature | Melting Point (Tm) | Glass Transition Temp (Tg) | Decomposition Temp |
Examples | PP, PE, PA (Nylon), POM, PEEK | ABS, PC, PS, PMMA (Acrylic) | Epoxy, Phenolic, Polyurethane |
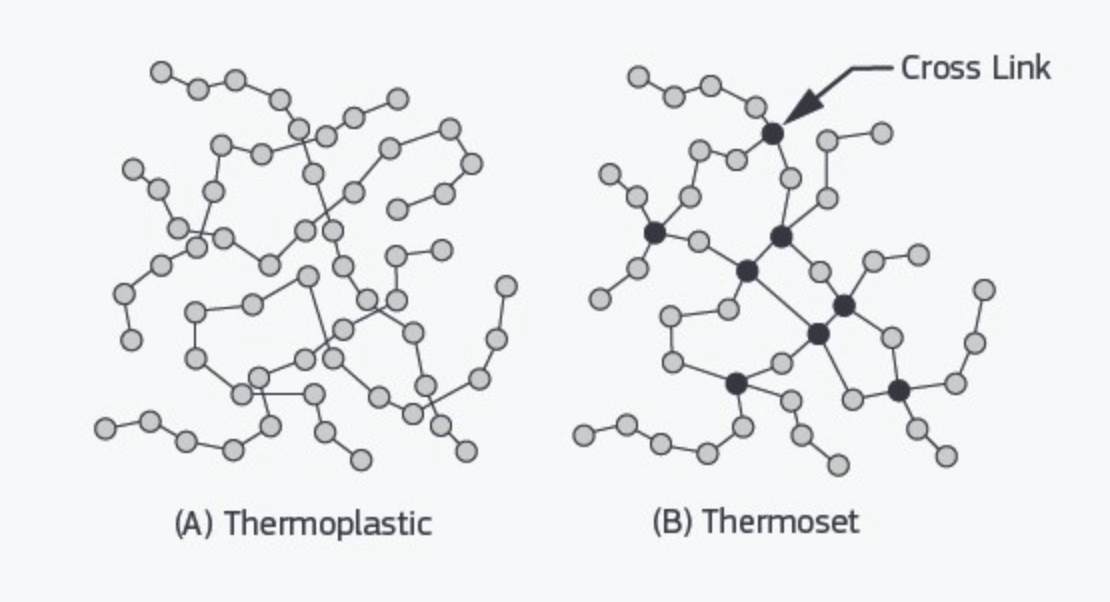
Thermoset vs Thermoplastic
source: researchgate.com
While the table above highlights the structural differences, the most practical distinction is reversibility.
Thermoplastics have a physical melting or softening point, which is reversible. They can be melted, reshaped, and remelted, making them suitable for recycling, injection molding, extrusion, and 3D printing.
Thermosets form permanent cross-links during curing. Once set, they do not melt; excessive heat causes decomposition instead of flow. This makes thermosets heat-resistant but impossible to remold using heat.
Thermoplastic: Semi-Crystalline vs Amorphous

source: becmaterials.com
Within thermoplastics, molecular structure defines melting behavior:
Semi-Crystalline plastics (e.g., Nylon, PP) have ordered crystalline regions. They exhibit a sharp melting point (Tm) and flow quickly once this temperature is reached. This behavior supports fast mold filling but often leads to higher shrinkage and opaque parts.
Amorphous plastics (e.g., ABS, PETG) lack ordered structures. They do not have a true melting point; instead, they gradually soften over a wide processing window—meaning they remain pliable across a broad temperature range. This behavior is governed by the Glass Transition Temperature (Tg) and generally results in transparent parts with low shrinkage.
To learn more about thermoplastic melting behavior, check out our detailed guides on PP, ABS, and PETG.
Plastic Melting Point Chart
The table below provides the thermal properties of common plastics, sorted by temperature from low to high.
Since amorphous plastics do not truly melt, the values listed refer to their Glass Transition Temperature (Tg)—the point where the material begins to soften. For semi-crystalline plastics, the listed value refers to the actual Melting Point (Tm).
(Note: These values are averages. Actual temperatures may vary depending on grade, fillers, and additives.)
Material | Full Name | Type | Thermal Transition | Temp (°C) | Temp (°F) |
|---|---|---|---|---|---|
Rigid PVC | Polyvinyl Chloride | Amorphous | Softening (Tg) | 80°C | 176°F |
PETG | Polyethylene Terephthalate Glycol | Amorphous | Softening (Tg) | 85°C | 185°F |
PS | Polystyrene | Amorphous | Softening (Tg) | 100°C | 212°F |
ASA | Acrylonitrile Styrene Acrylate | Amorphous | Softening (Tg) | 100°C | 212°F |
ABS | Acrylonitrile Butadiene Styrene | Amorphous | Softening (Tg) | 105°C | 221°F |
PMMA | Acrylic (Polymethyl Methacrylate) | Amorphous | Softening (Tg) | 105°C | 221°F |
LDPE | Low-Density Polyethylene | Semi-Crystalline | Melting (Tm) | 110°C | 230°F |
HDPE | High-Density Polyethylene | Semi-Crystalline | Melting (Tm) | 130°C | 266°F |
PC | Polycarbonate | Amorphous | Softening (Tg) | 147°C | 297°F |
PLA | Polylactic Acid | Semi-Crystalline | Melting (Tm) | 155°C | 311°F |
PP | Polypropylene | Semi-Crystalline | Melting (Tm) | 165°C | 329°F |
PA6 | Nylon 6 (Polyamide) | Semi-Crystalline | Melting (Tm) | 220°C | 428°F |
PEEK | Polyether Ether Ketone | Semi-Crystalline | Melting (Tm) | 343°C | 649°F |
Key Takeaways from the Chart:
Amorphous plastics (such as PVC, ABS, and PC) begin to soften at their glass transition temperature (Tg), but typically require higher temperatures to become fully flowable during processing.
Semi-crystalline plastics (such as HDPE, PP, and Nylon) remain solid until reaching their specific melting point (Tm), after which they liquefy rapidly.
PLA is a special case: it has a relatively low glass transition temperature (Tg ≈ 60 °C), but must reach its melting point (≈ 155 °C) to be properly molded or 3D printed.
Factors Influencing Plastic Melting Point
The melting point of a polymer is not a fixed universal constant; it is influenced by both the internal molecular architecture and external environmental conditions.
Several key factors determine the temperature at which a plastic transitions from solid to liquid:
Internal Factors
● Chemical Structure & Intermolecular Forces: The stronger the bond, the higher the melting point.
The strength of the "grip" between molecular chains determines the thermal energy required to separate them. Polymers with strong Hydrogen Bonding (e.g., Nylon) or Dipole-Dipole interactions (e.g., PVC) have higher melting points because their chains are more tightly bound together.
● Degree of Crystallinity: The higher the crystallinity, the sharper the melting point.
Crystallinity refers to how neatly the polymer chains are packed. Semi-crystalline plastics have highly ordered regions; as crystallinity increases, the melting point becomes higher and more precise. In contrast, amorphous plastics lack this order, resulting in a gradual softening range rather than a single temperature point.
● Chain Length & Branching: The longer and straighter the chains, the higher the melting point.
Molecular Weight: Longer chains have more surface area for entanglement and Van der Waals forces, generally increasing the melting point.
Branching: Highly branched chains (like LDPE) cannot pack together tightly. The more linear and "neat" the chains (like HDPE), the higher the melting point.
● Copolymerization: The more "mixed" the monomers, the lower the melting point.
By combining different monomers, manufacturers disrupt the regularity of a polymer chain. This irregularity hinders crystallization, a technique often used to intentionally lower the melting point for easier processing.
External Factors
● Additives & Plasticizers: The more plasticizers added, the lower the melting point.
Plasticizers act as "molecular lubricants" between polymer chains, increasing flexibility and distance between them. This significantly lowers both the glass transition temperature (Tg) and the melting point. A classic example is the difference between rigid PVC and flexible PVC.
● Moisture Content: The higher the moisture, the lower the melting point.
Certain plastics, particularly Polyamides (Nylon), are hygroscopic. Absorbed water molecules act as plasticizers, disrupting internal bonds and lowering the effective melting point and structural stiffness.
● Thermal History & Cooling Rate: The faster the cooling, the lower the melting point.
The "thermal memory" of a plastic matters. If a semi-crystalline plastic is cooled very rapidly (quenched), the chains lack the time to organize into crystals. This results in lower crystallinity and a modified (usually lower) melting profile during the next heating cycle.
Why Melting Point Matters in Material Selection
Choosing a material is a balancing act between performance, processability, and cost. While the melting point is a primary indicator of heat resistance, it also dictates how a project moves through the factory floor.
Practical Implications of Melting Point
To understand the practical implications of melting point, it helps to examine how it influences structural performance, manufacturing efficiency, and tooling requirements.
1.Structural Integrity in End Use
For high-stress environments such as automotive engine bays or aerospace electronics, the material must remain rigid under sustained operating temperatures. If service conditions approach the glass transition temperature (Tg) or melting temperature (Tm), the part may warp, creep, or fail entirely.
2.Manufacturing Efficiency
High-melting-point plastics (such as PEEK) require specialized heaters, insulated molds, and higher energy input. This increases the cycle time—the total time required to melt, inject, and cool a part—directly affecting production throughput and cost.
3.Tooling Requirements
Processing high-temperature materials typically demands hardened steel molds capable of withstanding repeated thermal cycling. In contrast, lower-temperature plastics may allow the use of aluminum tooling, which can be more economical for short production runs.
Melting Point vs. Heat Deflection Temperature (HDT)
Although the melting point (Tm) defines the temperature at which a crystalline polymer becomes a liquid, it is often not the limiting factor in real-world applications. The Heat Deflection Temperature (HDT) describes the temperature at which a plastic part deforms under a specified mechanical load.
In many cases, HDT is substantially lower than the melting point. A material may remain solid yet lose enough stiffness to compromise its function. For instance, a plastic enclosure may never reach its melting temperature during operation but can still deform or fail once the HDT is exceeded.
Consequently, melting point is primarily a processing parameter, whereas HDT is frequently the critical constraint for load-bearing parts.
Need help choosing the right plastic material for your application?
Talk to our experts for tailored material recommendations.
Why Melting Point Matters in Manufacturing Processes
Knowing the scientific melting point (Tm) or glass transition temperature (Tg) is just the starting point.
In a manufacturing environment, you act upon Processing Temperatures, which are typically set significantly higher than the material's physical melting point to account for viscosity, pressure, and flow rates.
Injection Molding: Melt and Mould Temperatures
● Melt Temperature (Barrel Temp): The temperature of the plastic as it enters the mold.
Rule of Thumb: Set 20°C - 50°C higher than Tm.
Reason: The plastic needs low viscosity to flow into thin cavities. If set exactly at Tm, the material may freeze before filling the mold (short shots).
● Mould Temperature: The temperature of the metal tool itself.
For Semi-Crystalline: A higher mould temp is often used for engineering plastics (like PEEK or Nylon) to promote crystallization and strength.
For Amorphous: A lower mould temp helps "freeze" the part quickly for faster cycle times.
3D Printing: Nozzle and Bed Temperatures
● Nozzle Temperature: Must be higher than Tm to ensure smooth extrusion.
Example: PLA melts at ~155°C, but prints at 190°C - 210°C.
● Bed Temperature: The Tg Rule.
Rule of Thumb: Set the heated bed at or slightly above the material's Tg.
Reason: Keeping base layers near Tg maintains a "rubbery" state, relieving internal stresses that lead to curling. Since ABS has a Tg of ~105°C, a bed temperature of 100°C–110°C is recommended.
Pro Tip: Incorrect bed temperatures lead to structural failures. Exploring effective solutions for 3D print warping can help maintain dimensional accuracy.
● Chamber Temperature: For high-temp industrial plastics like PEEK or Polycarbonate, a heated chamber is kept just below Tg to prevent cracking.
Extrusion
Extrusion temperatures change along the barrel:
Zone Temperatures: Cooler at the feed zone (preventing premature melting) and hotter towards the die (above Tm).
Die Temperature: Must be precisely controlled to ensure the plastic exits with a stable shape (controlling Die Swell).
Cooling Bath: The profile enters water cool enough to solidify the shape below Tm or Tg instantly.
Thermoforming
Thermoforming depends entirely on the Tg to rubbery-state range:
Target Temperature: Heat the sheet above Tg but below the melt flow point.
Rubbery State: The plastic is pliable enough to stretch without tearing, yet strong enough to hold cohesion.
Too Cold: Below Tg → sheet cracks.
Too Hot: Near Melt flow → sheet sags uncontrollably.
Example: ABS sheet is formed at ~140–150°C (well above its Tg of 105°C, but below its decomposition or flow temperature).
Want a quick idea of your project cost? Try our instant cost calculator.
Conclusion
Plastic melting point is a critical parameter, but it should never be evaluated in isolation. Different plastics soften or melt at different temperatures depending on their molecular structure.
While melting point and glass transition temperature define processing behavior, real-world performance is often limited by heat deflection temperature (HDT).
By understanding how these thermal properties translate into both manufacturing settings and end-use limits, engineers can select the right material for reliable, cost-effective performance.
FAQs
What is the melting point of plastic?
There is no single melting point for all plastics. Most common thermoplastics melt or process within a range of 170°C to 260°C (338°F to 500°F). However, high-performance materials like PEEK can have melting points as high as 343°C (649°F).
What temperature will plastic melt?
Plastic begins to lose its structural integrity at its Glass Transition Temperature (Tg) or its Melting Point (Tm). While engineering plastics can withstand high heat, many everyday plastics (like PLA or PET) can start to soften and warp at temperatures as low as 60°C (140°F).
Why do plastics have relatively low melting points?
Unlike metals, which are held together by strong metallic bonds, plastics are polymers held together by weaker intermolecular forces (such as Van der Waals forces). These bonds require much less thermal energy to break, allowing the molecular chains to slide past each other and liquefy at lower temperatures.
Will plastic melt at 95 degrees?
It depends on the scale and the material:
95°F (35°C): No. This is roughly body temperature; all industrial and consumer plastics remain completely solid.
95°C (203°F): It will deform, but typically not liquefy. Common plastics like LDPE, PVC, and PETG will become extremely soft, rubbery, and lose their shape (warp) at this temperature, but they usually require higher temperatures (110°C+) to turn into a flowable liquid.
What is the melting point of common plastics?
PVC: Softens (Glass Transition) at ~80°C (176°F).
Polypropylene (PP): Melts at 160°C – 170°C (320°F – 338°F).
Polycarbonate (PC): Softens at 147°C (297°F) and processes at 280°C+.
Nylon (PA6): Melts at 220°C (428°F).

