3D Printing in Medical Devices: 6 Key Applications

From prototypes to end-use parts: how 3D printing is applied to anatomical models, wearable devices, prosthetics, surgical guides, and medical implants.
Introduction
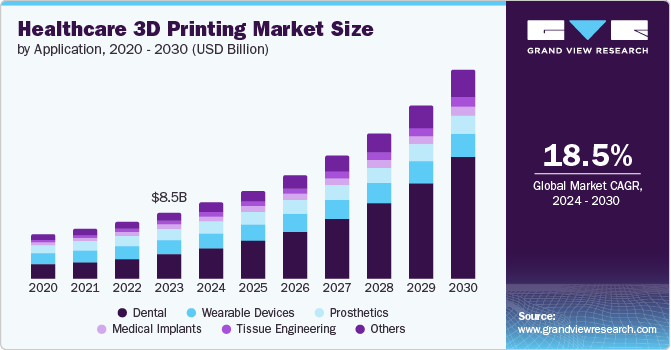
According to Grand View Research, the global healthcare 3D printing market reached USD 8.52 billion in 2023 and is forecast to grow to USD 27.29 billion by 2030, expanding at a CAGR of 18.5% from 2024 to 2030.
Healthcare 3D printing is a broad field covering multiple application areas, including medical implants, prosthetics, wearable devices, tissue engineering, dental products, and more.
This article focuses specifically on medical device applications, introducing six representative additive manufacturing scenarios:
Functional prototyping for R&D: Evaluate form, fit, and assembly to accelerate design iterations before expensive tooling.
Anatomical models: Scan-derived physical replicas for preoperative planning and surgical rehearsal.
Wearable devices: Custom-fit, skin-conforming housings and components generated directly from patient 3D scans.
Prosthetics: Lightweight, digitally customized structures (e.g., sockets) for optimal load distribution and daily comfort.
Surgical guides: Procedure-specific, sterilizable tools to ensure precise cutting and drilling alignment in the OR.
Medical implants: Patient-matched, biocompatible structures for complex reconstructions where off-the-shelf sizes fall short.
1. Functional Prototypes for Medical Device R&D
3D printing is widely used for rapid prototyping in medical device development, supporting iterative design, fit checks, and functional validation without relying on tooling or long supplier lead times.
Examples: handheld device housings, ergonomic handles, internal assembly mockups, fit-check components, and functional test parts.
Case 1: Abbott: Vascular Device Prototypes and Ergonomic Handles
At Abbott, 3D printing was already embedded in practical device development workflows as of 2015. The company describes using it for prototype devices, simulated clinical environments, and ergonomically shaped handles for vascular delivery systems.
For the Supera Peripheral Stent System, engineers printed and iterated on handle triggers, gears, and levers daily, compressing a development process that would otherwise take up to 10 months. Applications spanned vascular, diabetes, and diagnostics programs.

Case 2: University of Maryland: Automatic Vascular Access Device Prototype
At the University of Maryland, Baltimore, a mechanical engineer worked with a clinician in 2023 to develop a prototype for an Automatic Vascular Access Device (AVAD).
According to UM Ventures, the team used medical device prototyping and 3D printing resources to refine the design iteratively, and the resulting prototype supported key benchtop testing.

2. Anatomical Models for Clinical Planning
Not every medical device application of 3D printing becomes part of the final product. In many cases, the biggest value comes before a procedure begins.
Anatomical models help surgeons and care teams visualize patient-specific geometry, understand complex pathology, and plan difficult operations with more confidence.
This application is especially valuable when imaging alone does not provide enough spatial clarity. A physical model can improve communication, support rehearsal, and help teams anticipate procedural challenges before entering the operating room.
Examples: bone models, spinal models, vascular models, tumor models, and transplant planning models.
Case 1: Mayo Clinic: Life-Size Spinal Models for Scoliosis Planning
As of 2021, Mayo Clinic operates the largest point-of-care additive manufacturing facility in the United States, enabling surgical teams to move rapidly from patient imaging to life-size, patient-specific 3D-printed models.
For complex scoliosis cases, these models serve as presurgical planning tools that help surgeons understand anatomy that would otherwise require significant mental reconstruction from 2D scan slices.
The value here is not in the printed object as a clinical device, but in the spatial understanding it provides before the patient enters the operating room.
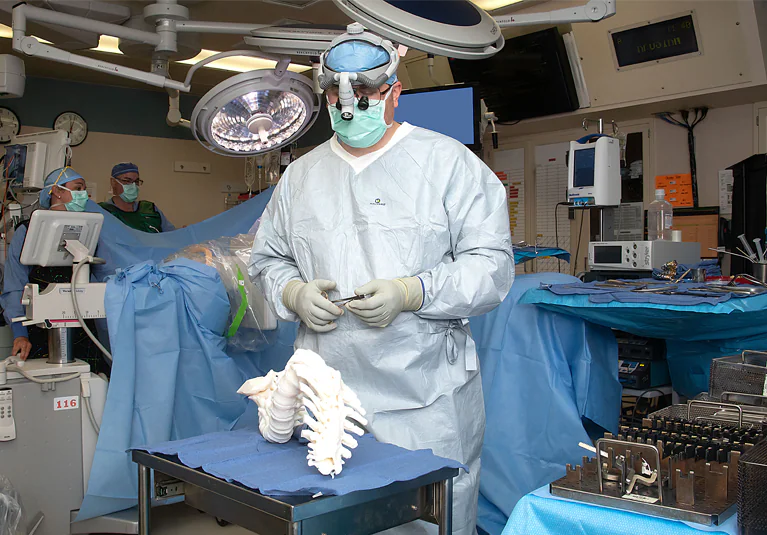
Case 2: Guy's and St Thomas': Pediatric Kidney Transplant Planning Models
In January 2016, Guy's and St Thomas' NHS Foundation Trust reported what was described as a world first: the use of 3D-printed anatomical models to support a kidney transplant from an adult donor into a two-year-old child.
Models of both the donor kidney and the child's abdomen were produced from CT and MRI data, allowing the surgical team to assess transplant feasibility and rehearse each step of the procedure before entering the operating room.
This case illustrates how 3D printing can bridge the gap between 2D imaging and the spatial understanding surgeons need when planning procedures where standard imaging alone is insufficient.

3. Wearable Medical Devices and Custom-Fit Components
3D printing enables patient-specific design for wearable medical devices, moving beyond generic prototyping. By conforming to individual anatomy, additive manufacturing creates lightweight, breathable components that offer improved comfort and fit compared to off-the-shelf alternatives.
This anatomy-driven approach ensures that body-worn devices, such as monitors and support structures, achieve the precise interface required for effective long-term usability.
Examples: custom-fit monitoring device housings, body-conforming sensor structures, braces, wearable support components, and scan-based external medical accessories.
Case 1: University of Arizona: Personalized Wireless Biosymbiotic Wearables
In 2021, engineers at the University of Arizona developed what they called "biosymbiotic devices," which are personalized, medical-grade wearables 3D-printed from body scans of individual wearers.
Because the devices are designed to operate continuously via wireless power transfer without ever needing to be removed for charging, precise anatomical fit is essential, making custom additive manufacturing central to the concept rather than incidental to it.
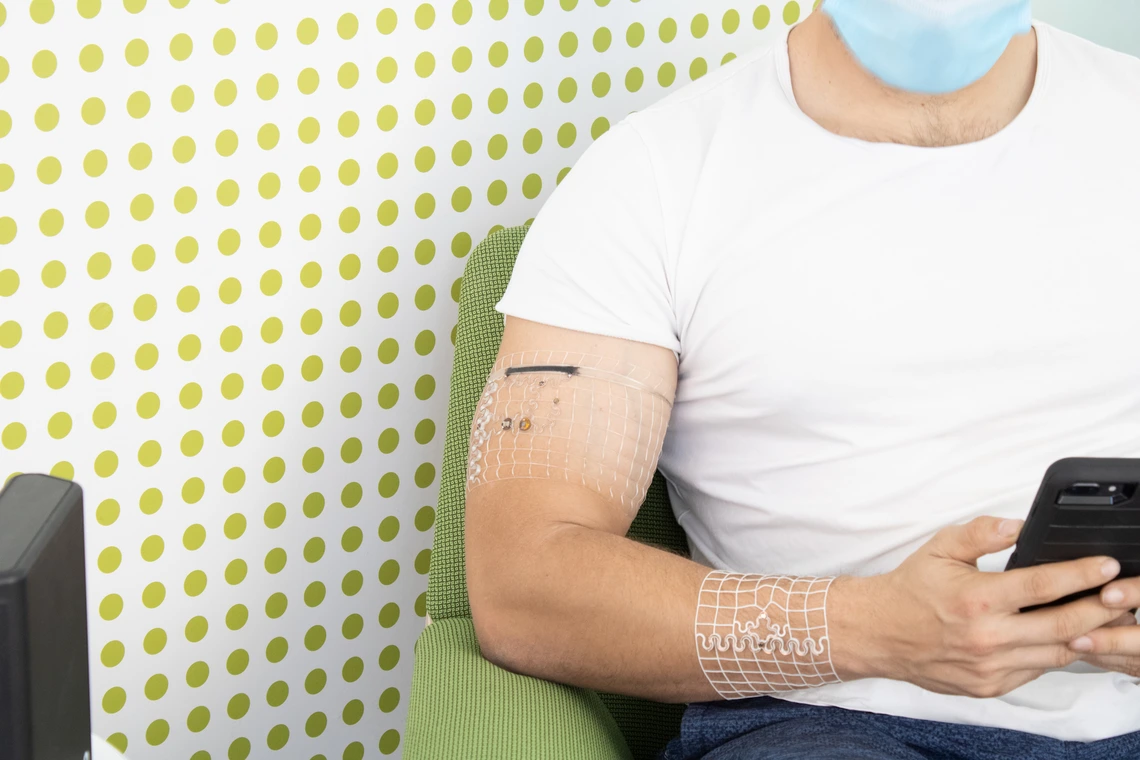
Case 2: University of Minnesota: 3D-Printed Light-Sensing Wearable for Lupus Monitoring
At the University of Minnesota, a research team spanning mechanical engineering and the Medical School developed a 3D-printed light-sensing device designed to interface directly with the skin. According to the university's release, the device provides real-time feedback correlating light exposure with disease flare-ups in people with lupus and other light-sensitive conditions.
The project illustrates how 3D printing can support the development of skin-interfaced medical sensing devices that combine custom form factors with functional electronics, even at the prototype and early clinical testing stage.

4. Prosthetic Devices and Components
3D printing provides a viable path for prosthetic devices requiring a balance of lightweight design and individual customization. While traditional methods often face cost barriers at low volumes, additive manufacturing allows for repeatable, tailored components that align with specific user needs.
These systems prioritize adaptation to patient anatomy and load distribution to ensure long-term comfort. The integration of digital design and 3D printing offers an effective approach for creating sockets, shells, and other structural elements that interface directly with the user.
Examples: prosthetic sockets, shells, upper-limb components, and lower-limb interface parts.
Case 1: Ottobock: MyFit TT 3D-Printed Prosthetic Socket
Ottobock brings this approach into day-to-day prosthetic care with its MyFit TT system. Using a scan-based workflow, the company produces definitive 3D-printed transtibial prosthetic sockets for daily use, replacing the conventional plaster casting process entirely.
The socket is printed using a powder-based nylon process inspired by the structure of human bone, giving it both the flexibility needed to don and doff the device and the robustness required for daily use.
The socket is a highly fit-sensitive component in a lower-limb prosthetic system. The ability to digitally capture residual limb geometry ensures that comfort and function are consistently achieved, while allowing adjustments to be reproduced more easily than with manual fabrication methods.

Case 2: Open Bionics: Hero Arm with Custom 3D-Printed Components
Open Bionics has developed the Hero Arm, described as the world's first clinically approved 3D-printed bionic arm, featuring multi-grip prosthetic hands.
This case illustrates how 3D printing can support both functional complexity and scalable production in external prosthetic systems, moving beyond prototyping into a clinically approved, commercially available product.

5. Surgical Guides & Patient-Specific Instrumentation
Surgical guides and patient-specific instruments represent a shift from external applications toward direct clinical contact during a procedure.
Unlike wearables or prosthetics that interface with the skin, these tools provide physical guidance for actions such as drilling or cutting by interacting directly with the operative site. This ensures that the surgical execution aligns with the digital plan through precise, patient-specific guidance.
Because these components make direct contact with tissue, bone, or teeth, they carry higher requirements for biocompatibility and sterilization compared to non-invasive devices. Even though their clinical use is temporary, the material must be safe for internal exposure to prevent adverse reactions.
For surgeons, this translates to improved positioning, more predictable preparation steps, and a closer match between the planned and executed procedure.
Examples: drill guides, cutting guides, positioning jigs, and dental implant guides.
Case 1: Mayo Clinic: Patient-Specific Instrumentation for Shoulder Arthroplasty
In shoulder arthroplasty, Mayo Clinic described in 2021 the use of patient-specific instrumentation derived from individual CT scans, including a 3D-printed surgical guide attached to the reamer for precise glenoid preparation.
This surgical guide allows surgeons to execute their preoperative plan in the operating room with greater accuracy, helping preserve glenoid bone and optimize implant positioning.

Case 2: Yale Orthopaedics: In-House 3D Surgical Guides for Deformity Correction
In 2024, Yale Orthopaedics reported its first fully in-house 3D surgical case, in which patient-specific jigs, surgical guides, and anatomical bone models were designed and printed on-site to support a distal radius malunion repair.
The procedure relied on a close surgeon-engineer collaboration: CT imaging was used to mirror the patient's healthy arm onto the affected side, simulate the osteotomy digitally, and finalize hardware placement before any incision was made.
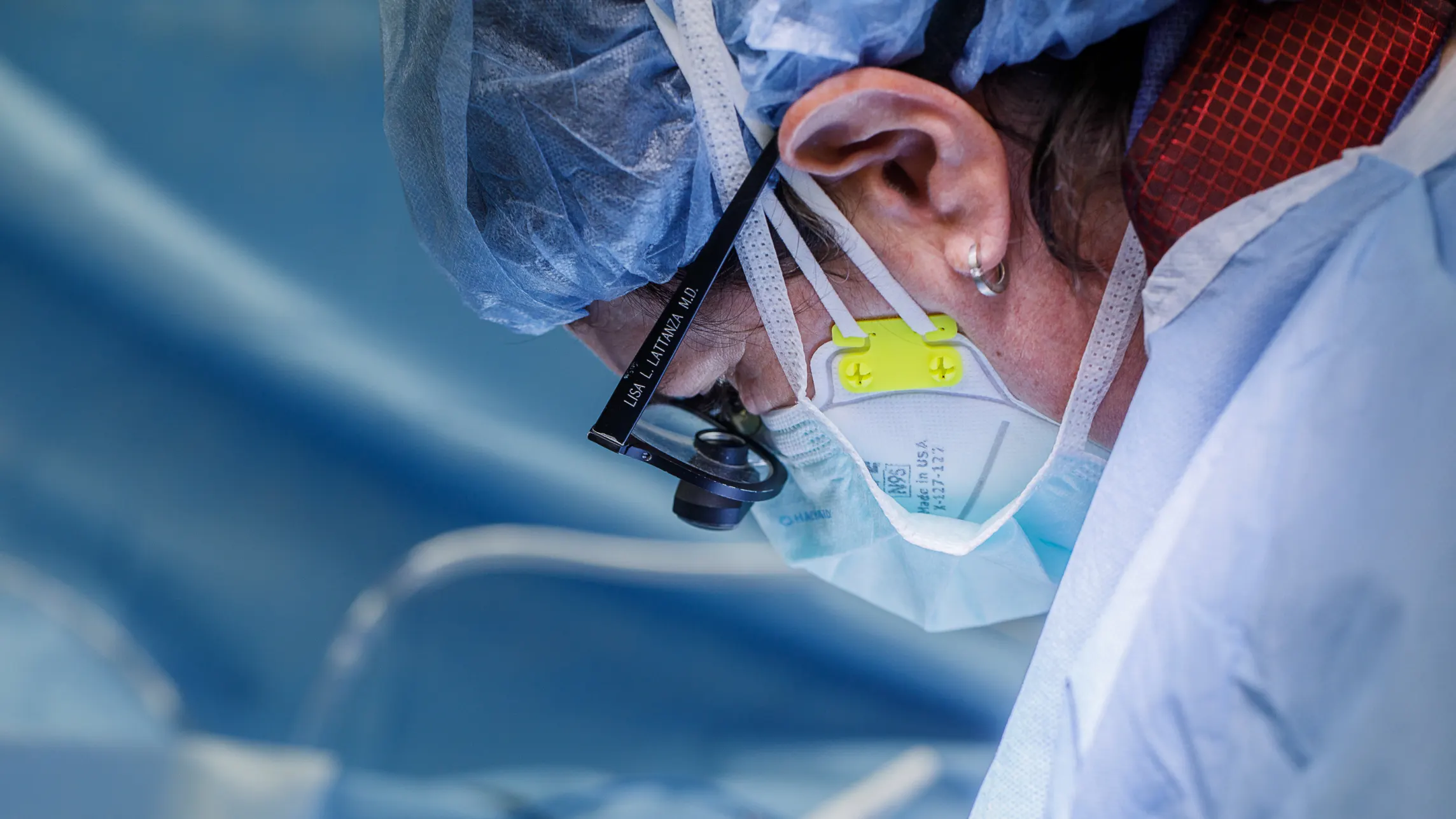
6. Patient-Specific Medical Implants and Implant Prototyping
Patient-specific implants involve a high level of clinical interaction as they are designed for permanent or long-term placement within the body. This application requires rigorous attention to biocompatibility, material validation, and traceability to meet stringent regulatory standards. Unlike temporary surgical tools, these devices must maintain their integrity and functionality while integrated into the patient’s physiological environment over time.
Additive manufacturing enables complex internal geometries and personalized surface structures that are often unattainable through conventional manufacturing methods.
Beyond the final device, this category also includes implant prototypes and fit-check models used during the development phase. These validation parts allow for precise testing of the device interface against the patient’s anatomy, ensuring the final implant meets the necessary requirements for safety and performance.
Examples: Cranial and CMF implants, spinal cages, orthopedic implant prototypes, and anatomy-matched validation parts.
Case 1: Hospital for Special Surgery: Patient-Specific Implants for Joint Reconstruction
In 2022, Hospital for Special Surgery announced the first surgeries completed using patient-specific 3D-printed implants produced at its on-site ProMade Point of Care Center, a collaboration with orthopedic manufacturer LimaCorporate.
The center allows surgeons and design engineers to collaborate directly before implants are printed on-site, reducing time to surgery by weeks compared to conventional international production workflows.
The first two cases involved a hip and a shoulder reconstruction, both selected for their anatomical complexity, illustrating how point-of-care additive manufacturing can provide viable solutions for patients with limited reconstructive options.
Source: news.hss.edu
Case 2: UF Health: Patient-Specific 3D-Printed Total Talus Implant
In 2023, UF Health reported the use of a patient-specific cobalt-chrome 3D-printed total talus implant in ankle replacement surgery.
The implant is dimensioned from a CT scan of the patient's own anatomy, replacing a bone that, due to its complex geometry and high cartilage coverage, is difficult to match through cadaveric grafts or conventional manufacturing.
Beyond fit, the clinical benefit is also functional: compared to traditional fusion procedures that require six or more weeks of non-weight-bearing recovery, patients with the 3D-printed implant were reported to be walking within one to two weeks.
Source: www.ortho.ufl.edu
Decision Matrix: Matching Applications with Tech & Materials
Selecting a 3D printing process involves evaluating several technical and regulatory factors beyond basic manufacturing speed. The decision depends on the functional requirements of the part, the nature of clinical contact, and the specific sterilization protocols involved.
Additionally, engineers must account for the required level of traceability and whether the project is in the prototyping stage or transitioning to low-volume production.
The following decision matrix provides a framework for aligning these application requirements with appropriate technologies and materials.
Application | Top Tech Choice | Go-To Material | The Core Value |
|---|---|---|---|
Functional Prototyping | Tough Resin | Delivers high-quality surface finishes and tight tolerances for verifying housings, ergonomics, and snap-fits. | |
Anatomical Models | Multi-Material Resin | Supports simultaneous printing of colors and textures to differentiate bone, soft tissue, and vascular networks. | |
Wearable Devices | TPU (Flexible) | Produces durable, lightweight, and skin-friendly structures that conform to specific patient geometry. | |
Prosthetics | PA12 (Nylon) | Provides the balance of lightweight design, tensile strength, and impact resistance required for load-bearing components. | |
Surgical Guides | Biocompatible Resin | Achieves high dimensional accuracy; specific resins are formulated to withstand high-temperature autoclave sterilization. | |
Orthopedic Implants | Ti6Al4V (Titanium) | Creates patient-specific structures with engineered porosity to mimic bone modulus and promote osseointegration. | |
Spinal Implants | PEEK | Offers an elastic modulus similar to human bone to prevent stress shielding and provides radiolucency for post-op imaging. |
Want to determine the costs for your next 3D-printed medical device?
Partnering with an ISO 13485-Certified Manufacturer
Transitioning a medical device from early R&D to clinical application requires more than just advanced 3D printers. It demands strict regulatory compliance and quality assurance.
Unionfab holds an ISO 13485 certification, establishing a quality management system built specifically for the stringent requirements of medical device manufacturing. Whether you are scaling up surgical tools, wearable components, or anatomical models, partnering with us ensures:
End-to-End Traceability: Complete material and process documentation for regulatory confidence.
Rigorous Process Control: Repeatable precision that general-purpose prototyping labs simply cannot match.
Seamless Scaling: Transition smoothly from single functional prototypes to low-volume, high-quality production.
Ready to place your first order? New customers get 10% off.
FAQs
How is 3D printing used in medical device manufacturing?
3D printing is used across several stages of medical device manufacturing. It supports early R&D prototypes, wearable device components, prosthetic parts, anatomical models for surgical planning, patient-specific guides, and selected implant-related workflows.
What are 3D printed medical implants?
3D printed medical implants are implantable parts manufactured through additive processes to match a clinical design requirement or, in some cases, a patient's specific anatomy. They are often discussed in areas such as orthopedic, cranial, CMF, or spinal reconstruction.
What is point-of-care 3D printing in medical devices?
Point-of-care 3D printing in medical devices refers to additive manufacturing performed within or near a hospital or clinical setting to support patient-specific care. It is commonly used for anatomical models, surgical planning aids, and selected procedural tools.

